|
Since there is very little difference in the molar masses, the entropy decrease can only be attributed to the increase in the coulombic attraction between the ions as we move from the singly charged ions Na + and F – through the doubly charged ions Mg 2+ and O 2–, to the triply charged ions A1 3+ and N 3–. Which are isoelectronic with sodium fluoride. A good example is the three solid compounds Strength of Bonding Another trend in entropy, most noticeable in the case of solids, is the decrease in the entropy as the forces between the atoms, molecules, or ions increases. In consequence there are more ways in which the energy can be distributed among the molecules. the entropy of a pure substance at 298 K and 1 atm pressure). Standard molar entropies are listed for a reference temperature (like 298 K) and 1 atm pressure (i.e. The more atoms there are in a molecule, the more ways the molecule can change its shape by vibrating. Standard Molar Entropy, S 0 The entropy of a substance has an absolute value of 0 entropy at 0 K. Since T is the absolute (Kelvin) temperature, it can only have positive values. However, we still find an increase of entropy with complexity when we compare molecules of very similar masses: The extent of a process, as reflected in the arithmetic sign of its standard free energy change, is then determined by the signs of the enthalpy and entropy changes and, in some cases, the absolute temperature. To some extent this is due to the mass since on the whole more complex molecules are heavier than simpler ones. Molecular Complexity A second clear trend in the table is the higher molar entropy of substances with more complex molecules. When 1 mol of ice melts, for example, its entropy increases by 22 J K –1, while on boiling the entropy increase is 110 J K –1. When a liquid vaporizes, the restrictions on the molecules’ ability to move around are relaxed almost completely and a further and larger increase in the entropy occurs. 
Thus when a solid melts, the molar entropy of the substance increases. 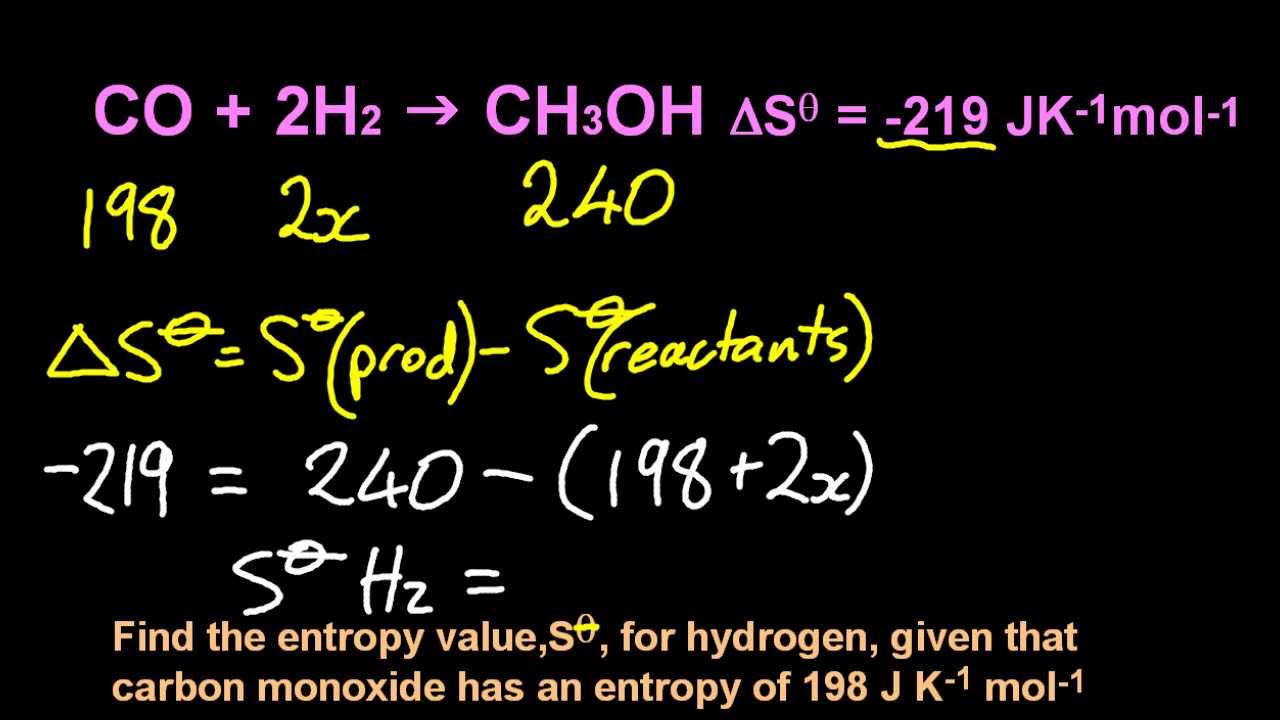
In a solid, the molecules are only capable of restricted vibrations around a fixed point, but when a solid melts, the molecules, though still hampered by their mutual attraction, are much freer to move around. Solids, Liquids, and Gases Perhaps the most obvious feature of the table of molecular entropies is a general increase in the molar entropy as we move from solids to liquids to gases.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
